t is thought that PRRS virus has a strongly restricted tropism for only those sub-populations of macrophages found in lungs, placenta and lymphoid organs that display two specific glycoproteins: Sialoadhesin and CD163. It seems that the virus cannot replicate in monocytes in circulation or resident dendritic cells in the lungs. However, recent studies point out that PRRS virus could replicate in other subtype of macrophages or dendritic cells. Initially, virus replication occurs in local permissive macrophages and then spreads to the lungs and lymphoid organs, such as the tonsils and lymph nodes, and less consistently, to other tissues. The virus can be detected in blood (viraemia) within hours (12-24h). The length of viraemia is strongly influenced by the strain and the animal’s age. Thus, viraemia may extend for several weeks in piglets, whereas in adult pigs it may last for only a few days.
In lungs, the predominant differentiated cells that support PRRS virus replication are pulmonary alveolar macrophages and pulmonary intravascular macrophages. The virus compromises their functions and provokes an inflammatory cell infiltration resulting in interstitial pneumonia which, in conjunction with enlarged lymph nodes, is the common lesion in a PRRS virus infection. Microscopically, lesions can be also observed in the uterus of sows and in the testicles of boars. Clinical disease and lesions due to the PRRS virus infection occur by a variety of mechanisms that include apoptosis of infected and noninfected macrophages, induction of polyclonal B-cell activation and induction of inflammatory cytokines.
Among other causes, the severity of clinical signs strongly depends on the infecting strain, the presence of concurrent infections, the host’s immune status, and the age of the pig.
In viraemic pregnant sows, it is known that PRRS virus can infect foetuses, reaching high titers in them. The virus crosses the placental barrier most efficiently in late gestation (after day 70, approximately). Recent studies have indicated that the endometrium and the placenta are involved in the PRRS virus passage from sow to foetus and that foetal death is caused by virus replication in the endometrial and placental tissues. As a result, PRRS virus can cause abortion or affect the viability of a variable percentage of the foetuses.
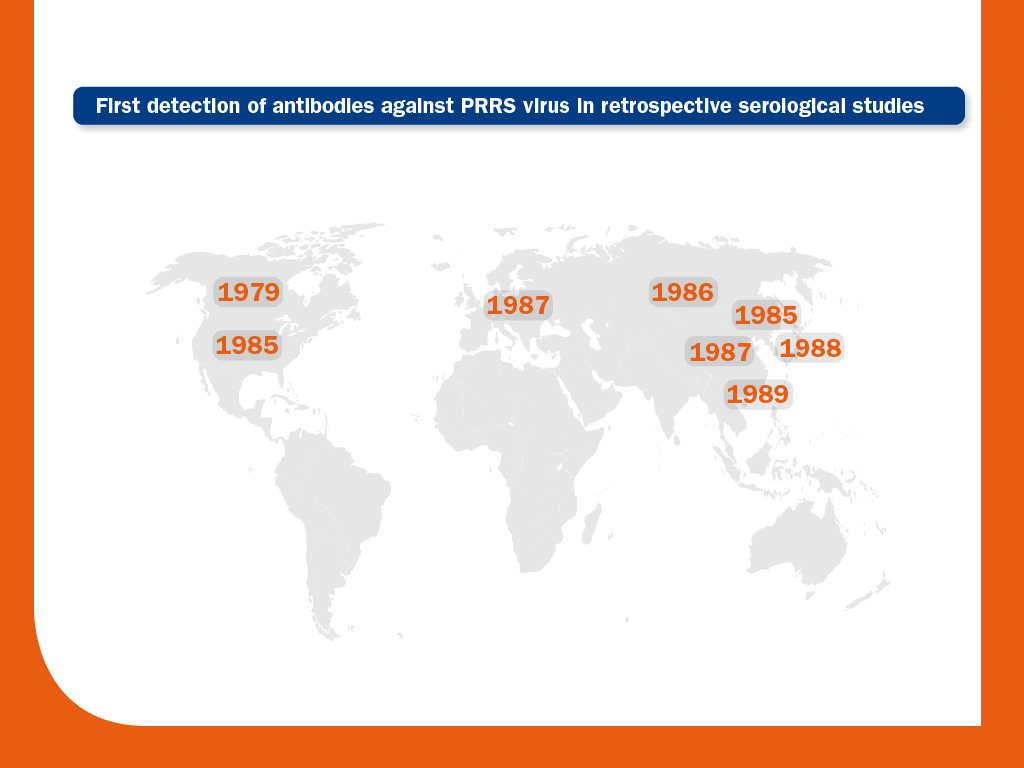
The earliest evidence of PRRS virus comes from retrospective serological studies; antibodies have been detected in serum as early as 1979 (Canada). In the U.S., retrospective analysis found no evidence of infection until 1985. Its prevalence then increased rapidly in North-America until 1988-89, when clinical outbreaks were first reported. There was probably a similar pattern in Europe. In Asia, the first evidence of antibodies has been retrospectively documented in serum from 1985 (South Korea). These results suggest that PRRS virus had probably been circulating in domestic pigs for several years before the first outbreaks were reported.
Curiously, we are facing a disease caused by one virus with two different strains (genotypes) that appeared almost simultaneously on two different continents. The explanation behind this phenomenon is unclear. Nevertheless, two main hypotheses for the uncertain origin of the virus have been postulated:
- Origin close in time. Origin just one decade before the virus emerged in domestic pigs. This hypothesis would imply that two different genotypes appeared in a very short period of time and that the virus should have an excessively high mutation rate, even for PRRS virus.
- Origin far in time. The genotypes would have developed independently. Using ORF3 as a molecular clock, it has been estimated that PRRS virus could have originated in 1880±15 years. Thus, genotypes evolved over decades in two different and isolated environments before they emerged in pigs. This estimation is compatible with the hypothesis postulated by Plagemann (2003). According to this author, PRRS virus originated from an ancestor similar to Lactate dehydrogenase-elevating virus (another Arterivirus) in mice from Eastern Europe. This ancestor jumped from mice to wild boars and then the movement of these animals to a hunting reserve in USA made it possible for the ancestor to spread in North-America. Since then, the virus has evolved in parallel on both continents. Finally, changes in pork production would have facilitated the emergence of the disease. In spite of the congruency between this hypothesis and the molecular clock analysis, there is no evidence for the existence of a common ancestor in mice or wild boars.
© Laboratorios Hipra, S.A. 2015. All Rights Reserved.
No part of this website or any of its contents may be reproduced, copied, modified or adapted, without the prior written consent of HIPRA.
- Bautista EM, Meulenberg JJ, Choi CS, Molitor TW. Structural polypeptides of the American (VR-2332) strain of porcine reproductive and respiratory syndrome virus. Arch Virol. 1996:1357-65.
- Benfield DA, Nelson E, Collins JE, Harris L, Hennings JC, Shaw DP, Goyal SM, McCullough S, Morrison RB, Joo HS, Gorcyca D, Chladek D. Characterization of swine infertility and respiratory syndrome (SIRS) virus (isolate ATCC VR-2332). J Vet Diagn Invest. 1992. 4, 127-133.
- Benfield D, Nelson J, Rossow K, Nelson C, Steffen M, Rowland R. Diagnosis of persistent or prolonged porcine reproductive and respiratory syndrome virus infections Vet Res. 2000, 31:71.
- Brockmeier SL, Halbur PG, Thacker EL. Porcine respiratory disease complex. In KA Brogden, JM Guthmiller, eds, Polymicrobial Diseases. Washington, DC: ASM Press. 2002, 231-58.
- Calzada-Nova G, Schnitzlein W, Husmann R, Zuckermann FA. Characterization of the cytokine and maturation responses of pure populations of porcine plasmacytoid dendritic cells to porcine viruses and toll-like receptor agonists. Vet Immunol Immunopathol. 2010, 135:20-33.
- Chen WY, Schniztlein WM, Calzada-Nova G, Zuckermann FA. Genotype 2 strains of porcine reproductive and respiratory syndrome virus dysregulate alveolar macrophage cytokine production via the unfolded protein response. J Virol.2017, pii: JVI.01251-17.
- Christopher-Hennings J, Nelson EA, Hines RJ, Nelson JK, Swenson SL, Zimmerman JJ, Chase CL, Yaeger MJ, Benfield DA. Persistence of porcine reproductive and respiratory syndrome virus in serum and semen of adult boars. J Vet Diagn Invest. 1995, 7:456-64.
- Cortey M, Díaz I, Martín-Valls GE, Mateu E. Next-generation sequencing as a tool for the study of Porcine reproductive and respiratory syndrome virus (PRRSV) macro- and micro- molecular epidemiology. Vet Microbiol. 2017. doi: 10.1016/j.vetmic.2017.02.002.
- Darwich L, Díaz I, Mateu E. Certainties, doubts and hypotheses in porcine reproductive and respiratory syndrome virus immunobiology. Virus Res. 2010, 154:123-32.
- Díaz I, Darwich L, Pappaterra G, Pujols J, Mateu E. Different European-type vaccines against porcine reproductive and respiratory syndrome virus have different immunological properties and confer different protection to pigs. Virology. 2006, 351:249-59.
- Díaz I, Gimeno M, Darwich L, Navarro N, Kuzemtseva L, López S, Galindo I, Segalés J, Martín M, Pujols J, Mateu E. Characterization of homologous and heterologous adaptive immune responses in porcine reproductive and respiratory syndrome virus infection. Vet Res. 2012, 19:43:30.
- García-Nicolás O, Quereda JJ, Gómez-Laguna J, Salguero FJ, Carrasco L, Ramis G, Pallarés FJ. Cytokines transcript levels in lung and lymphoid organs during genotype 1 Porcine Reproductive and Respiratory Syndrome Virus (PRRSV) infection. Vet Immunol Immunopathol. 2014, 160:26-40.
- Gimeno M, Darwich L, Diaz I, de la Torre E, Pujols J, Martín M, Inumaru S, Cano E, Domingo M, Montoya M, Mateu E. Cytokine profiles and phenotype regulation of antigen presenting cells by genotype-I porcine reproductive and respiratory syndrome virus isolates. Vet Res. 2011, 42:9.
- Gómez-Laguna J, Salguero FJ, Pallarés FJ, Carrasco L. Immunopathogenesis of porcine reproductive and respiratory syndrome in the respiratory tract of pigs. Vet J. 2013, 195:148-55.
- Haynes JS, Halbur PG, Sirinarumitr T, Paul PS, Meng XJ, Huffman EL. Temporal and morphologic characterization of the distribution of porcine reproductive and respiratory syndrome virus (PRRSV) by in situ hybridization in pigs infected with isolates of PRRSV that differ in virulence. Vet Pathol. 1997, 34:39-43.
- Hill H. Overview and history of Mystery Swine Disease (swine infertility/respiratory syndrome). Proceedings of the Mystery Swine Disease Committee Meeting, Livestock Conversation Institute, Denver, CO. 1990, 29–31.
- Holtkamp DJ, Polson DD, Torremorrell M. Terminology for classifying swine herds by porcine reproductive and respiratory status. J Swine Health and Prod. 2011, 19:44-56.
- Horter DC, Pogranichniy RM, Chang CC, Evans RB, Yoon KJ, Zimmerman JJ. Characterization of the carrier state in porcine reproductive and respiratory syndrome virus infection. Vet Microbiol. 2002, 86:213-28.
- Loula T. Mystery pig disease. Agri-practice. 1991, 12:23–34.
- Lunney JK, Benfield DA, Rowland RR. Porcine reproductive and respiratory syndrome virus: an update on an emerging and re-emerging viral disease of swine. Virus Res. 2010, 154:1-6.
- Mateu E, Tello M, Coll A, Casal J, Martín M. Comparison of three ELISAs for the diagnosis of porcine reproductive and respiratory syndrome. Vet Rec. 2006, 159:717-8.
- Mateu E, Diaz I. The challenge of PRRS immunology. Vet J. 2008, 177:345-51.
- Meier WA, Galeota J, Osorio FA, Husmann RJ, Schnitzlein WM, Zuckermann FA. Gradual development of the interferon-gamma response of swine to porcine reproductive and respiratory syndrome virus infection or vaccination. Virology. 2003, 309:18-31.
- Nelson EA, Christopher-Hennings J, Benfield DA. Serum immune responses to the proteins of porcine reproductive and respiratory syndrome (PRRS) virus. J Vet Diagn Invest. 1994, 6:410-5.
- Appendices IV and V of the REPORT OF THE OIE AD HOC GROUP ON PORCINE REPRODUCTIVE RESPIRATORY SYNDROME Paris, 9 – 11 June 2008 http://www.oie.int/fileadmin/Home/eng/Our_scientific_expertise/docs/pdf/PRRS_guide_web_bulletin.pdf.
- Rodríguez-Gómez IM, Gómez-Laguna J, Carrasco L. Impact of PRRSV on activation and viability of antigen presenting cells. World J Virol. 2013, 2:146-51.
- Rovira A, Cano JP, Muñoz-Zanzi C. Feasibility of pooled-sample testing for the detection of porcine reproductive and respiratory syndrome virus antibodies on serum samples by ELISA. Vet Microbiol. 2008, 130:60-8.
- Rovira A, Clement T, Christopher-Hennings J, Thompson B, Engle M, Reicks D, Muñoz-Zanzi C. Evaluation of the sensitivity of reverse-transcription polymerase chain reaction to detect porcine reproductive and respiratory syndrome virus on individual and pooled samples from boars. J Vet Diagn Invest. 2007, 19:502-9.
- Rovira A, Reicks D, Muñoz-Zanzi C. Evaluation of surveillance protocols for detecting porcine reproductive and respiratory syndrome virus infection in boar studs by simulation modeling. J Vet Diagn Invest. 2007, 19:492-501.
- Salguero FJ, Frossard JP, Rebel JM, Stadejek T, Morgan SB, Graham SP, Steinbach F. Host-pathogen interactions during porcine reproductive and respiratory syndrome virus 1 infection of piglets. Virus Res. 2015, 202:135-43.
- Segalés J, Domingo M, Balasch M, Solano GI, Pijoan C. Ultrastructural study of porcine alveolar macrophages infected in vitro with porcine reproductive and respiratory syndrome (PRRS) virus, with and without Haemophilus parasuis. J Comp Pathol. 1998, 118:231-43.
- Sur JH, Cooper VL, Galeota JA, Hesse RA, Doster AR, Osorio FA. In vivo detection of porcine reproductive and respiratory syndrome virus RNA by in situ hybridization at different times postinfection. J Clin Microbiol. 1996, 34:2280-6.
- Terpstra C, Wensvoorst G, Pol JMA. Experimental reproduction of porcine epidemic abortion and respiratory syndrome (mystery swine disease) by infection with Lelystad virus: Koch’s postulates fulfilled. Vet Q. 1991, 13:131–36.
- Tian K, Yu X, Zhao T, Feng Y, Cao Z, Wang C, Hu Y, Chen X, Hu D, Tian X, Liu D, Zhang S, Deng X, Ding Y, Yang L, Zhang Y, Xiao H, Qiao M, Wang B, Hou L, Wang X, Yang X, Kang L, Sun M, Jin P, Wang S, Kitamura Y, Yan J, Gao GF. Emergence of fatal PRRSV variants: unparalleled outbreaks of atypical PRRS in China and molecular dissection of the unique hallmark. PLoS One. 200, 2:e526.
- Tong GZ, Zhou YJ, Hao XF, Tian ZJ, An TQ, Qiu HJ. Highly pathogenic porcine reproductive and respiratory syndrome, China. Emerg Infect Dis. 2007, 13:1434-6.
Van Alstine WG, Popielarczyk M, Albregts SR. Effect of formalin fixation on the immunohistochemical detection of PRRS virus antigen in experimentally and naturally infected pigs. J Vet Diagn Invest. 2002, 14:504-7. - Vézina SA, Loemba H, Fournier M, Dea S, Archambault D. Antibody production and blastogenic response in pigs experimentally infected with porcine reproductive and respiratory syndrome virus. Can J Vet Res. 1996, 60:94-9.
- Wensvoort G, Terpstra C, Pol JMA, Lask EA, Bloemraad M, de Kluyver EP, Kragten C, van Butten L, den Besten A, Wagenaar F, Broekhuijsen JM, Moonen PJM, Zetstra T, de Boer EA, Tibben AhJ, de Jong MF, van’r Veld P, Groenland GJR, van Gennep JA, Voets MTh, Verheijden JHM, Braamkamp J. Mystery swine disease in the Netherlands: the isolation of Lelystad virus. Vet Q. 1991, 13:121–30.
- Wills RW, Doster AR, Galeota JA, Sur JH, Osorio FA. Duration of infection and proportion of pigs persistently infected with porcine reproductive and respiratory syndrome virus. J Clin Microbiol. 2003, 41:58-62.
- Zimmerman JJ, Benfield DA, Dee SA, Murtaugh MP, Stadejek T, Stevenson GW, Torremorell M. Porcine reproductive and respiratory syndrome virus (porcine arterivirus). In: 10th ed. Diseases of swine, Ed. Wiley-Blackwell. 2012, 31:463-86.